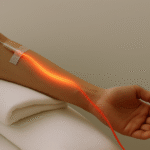
What Is It?
This therapy merges two key elements:
-
Methylene Blue (MB): A synthetic dye that becomes highly reactive when exposed to red light. It concentrates in areas of inflammation, infection, and mitochondrial dysfunction.
-
Red IV Laser Light: Delivered via a fiber-optic line placed into a vein (similar to an IV), this light therapy emits a precise 660 nm wavelength—ideal for activating Methylene Blue in the bloodstream.
When combined, they trigger photodynamic activity that can modulate the immune system, reduce pathogens, and energize the body from within.
How It Works
-
Step 1: Methylene Blue Ingestion
-
A liposomal form of MB is taken orally, usually 2 hours before the IV laser.
-
If other IVs (like ozone) are scheduled first, time the MB so that red light is administered 2–3 hours later.
-
-
Step 2: IV Red Laser Activation
-
A sterile fiber-optic catheter is inserted into a vein.
-
660 nm red light flows through the bloodstream, activating the MB wherever it has concentrated.
-
-
Step 3: Cellular Response
-
Activation generates singlet oxygen and ROS (reactive oxygen species).
-
These agents selectively target infected, inflamed, or dysfunctional cells.
-
Mitochondria may receive a temporary boost, enhancing energy production.
-
Blood flow and oxygenation often improve—a benefit known as enhanced rheology.
-
| Wavelength | Color | Function |
|---|---|---|
| 660 nm | Red | Activates Methylene Blue efficiently and safely |
What Conditions Might Benefit?
✅ Bioregulatory & Integrative Clinics
-
Chronic infections: Lyme disease, EBV, mold illness
-
Chronic fatigue and fibromyalgia
-
Immune dysregulation
✅ Longevity & Biohacking
-
Mitochondrial support
-
Synergy with NAD+, CoQ10, or ketones
-
Enhancing metabolism and cognitive clarity
✅ Post-Viral & Recovery Protocols
-
Long-haul viral symptoms
-
Immune rebalancing
-
Cellular detox and oxygenation
Why Methylene Blue?
Methylene Blue isn’t just a dye—it’s a molecule with a mission:
-
Selective uptake: Accumulates in stressed, infected, or energy-depleted cells
-
Low-light activation: Easily activated with red light—no heat or damage
-
Dual roles: Works as both a redox balancer and ROS generator
-
Safe profile: Generally well-tolerated at therapeutic dosages
Methylene Blue + IV Red Light therapy is more than just high-tech wellness—it’s mitochondrial medicine in action. With roots in science and results showing promise in energy restoration, immune modulation, and pathogen targeting, it’s no wonder this therapy is gaining traction in integrative settings.
Whether you’re recovering from chronic illness or just looking for a next-level mitochondrial tune-up, this dynamic duo might be worth exploring.